 |
Michael
Hooker Microscopy
Facility
(MHMF.ORG) |
 |
.gif) Leica
DMIRB
Inverted Microscope with Color & B/W Cooled Digital
Cameras
Leica
DMIRB
Inverted Microscope with Color & B/W Cooled Digital
Cameras
Location: 6129 Thurston Bowles
Notices
- If you are the last user of the day, especially later in the day,
please turn off the mercury arc lamp when you are finished.
- Please do not start mercury arc lamp if lamp housing is warm. It
must be cool! (In order to avoid damage or explosion the mercury arc
lamps must cool for ~30 minutes before being restarted.)
- If you plan to use this microscope for more than ~30 minutes at a time
please book ahead at the
Leica Inverted microscope booking page.
- Camera ports:
- The left side port transmits 100% or 0% of the light with a
magnification of 0.63x to a B/W high sensitivity Photmetrics HQ2 or Hamamatsu Orca ER CCD camera.
- The top trinocular port transmits 100% or 50% or 0% of light to a Q-Imaging MicroPublisher CCD color camera and has a 1x magnification.
Its suggested to leave it in the 50%/50% position.
- Fluorescence filter cube for far-red emitting fluorophores (e.g. Cy5, Alexa 633)
is stored in the plastic tub on the shelf above the scope.
- Clean install of the system (April 25, 2014) - All
users must setup C-Imaging profiles again. See Michael for
assistance or see here for
online instructions.
Contents
Introduction
The Leica microscope is a standard inverted microscope
set up for transmitted light, differential interference contrast (DIC or Nomarski) and
fluorescence microscopy. Images may be viewed by eye or viewed and saved
to computer with either of 2 cameras. For transmitted light and very
bright fluorescence imaging a chilled color camera is mounted on top of the
trinocular and can be used to capture high resolution digital images. For
dimly fluorescent samples or photometric quantification, a cooled B/W CCD camera (an
HQ2) made
by Photometrics is attached to the side port on the left of the microscope.
DIC images may also be captured using either camera, although the color camera
can capture with higher resolution. Darkfield is also available forthe 40x
& 63x oil immersion objectives. The specific hardware attached to this
microscopy is listed below.
The Equipment
The Microscope:
- Leica inverted DMIRB microscope
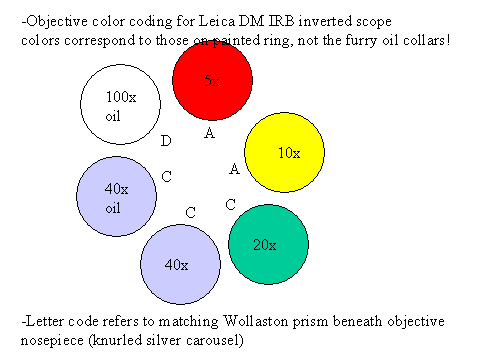
- Objectives:
|
.gif) |
| Objectives |
| Mag. |
NA |
type |
WD |
Adjustable Collar |
cover slip |
Immersion |
part no. |
Objective Wollaston *** |
Condenser Wollaston |
| 5X * |
0.11 |
Plan |
~9 mm |
- |
#1.5 |
air |
|
20 |
B1 |
| 10x * |
0.3 |
Fluor |
6 mm |
- |
#1.5 |
air |
|
20 |
B1 |
| 20x |
0.4 |
|
long |
corr |
0 - 2.0 mm |
air |
|
20 |
C or D |
| 40x |
0.55 |
|
long |
corr |
0 - 2.0 mm |
air |
|
40 |
C or D |
| 40x ** |
1.0 to 0.5 |
Plan Fluor |
240 um |
aperture |
#1.5 |
oil |
|
40 |
C or D |
| 100x ** |
1.4 to 0.7 |
PlanApo |
90 um |
aperture |
#1.5 |
oil |
|
100 |
C or D |
Use only Leica immersion oil with oil immersion objectives
* These
objectives prefer a number #1.5 (~170 um thick) glass cover slip.
** Suitable only for
imaging though cover slips due to small working distance
*** When not using DIC,
the objective Wollaston should be in the blank (H) position
corr - cover slip adjustment should be set for maximum clarity with fine
high contrast objects or set the dot on the dial on objective to the
corresponding objective thickness. e.g. for #1.5 (170um) cover slips dot should
be at 0.2 (you'll have to interpolate), or e.g. standard culture dishes (1.0 mm
thickness) set dot to 1.0
- Condenser:
- Type S1 0.9 NA 2 mm working distance (small conical nose lens,
screw on/off)
- Type S23 0.5 NA 23 mm working distance (large cylindrical nose lens,
screw on/off)
- Type S70 0.? NA 70 mm working distance (unscrew nose lens
from bottom of S23)
- Changing between S1, S23 & S70 requires a 3 mm hex wrench so unlock
the condensor assemble and slide the top to match the S number on the stem
of the microscope. Please keep a hand under the condensor when you
do this in order to prevent the assembly falling onto the objective.
- Differential Interference Contrast (Nomarski) is available for some of the
objectives
- Illumination:
- Transmitted light - 100 Watt Halogen (Tungsten filament) lamp with neutral density filters and a
color temperature correction filter
- Fluorescence - 100 watt Mercury arc lamp (HBO)
- Dichroic Filters for Fluorescence -
- Dapi (31000,
Chroma) ex=360/40 di=400lp em=460/50
- Texas Red (31004,
Chroma) ex=560/40 di=595lp em=630/60
- Fitc (41001,
Chroma) ex=480/40 di=505lp em=535/50
- Cy5 (41008, Chroma) ex=620/60 di=660lp em=700/75
- GFP (31019,
Chroma) ex=425/40 di=460lp em=505/40
(important:- this filter is not suitable for
the more common eGFP)
- eGFP Narrow Band (31026,
Chroma) ex=480/20 di=505lp em=520/20
(good for minimizing autofluorescence)
- Triple Dichroic combined Dapi, FITC, Texas Red (61002
Chroma) (note that
this filter set can have high cross talk between the fluorescent channels)
- Note 1: The GFP cubes are usually in the plastic tube on the shelf above
the microscope
- Note 2: The numbered positions of the cubes in the carrousel are
sometime
changed
- Fluorescent
proteins spectra - Clontech
Cameras:
- Cooled OrcaER (b/w 1.3 mega pixels)
- Acquisition through C-Imaging only
- Cooled Photometrics Cool Snap HQ2 (b/w 1.3 mega pixels)
- Acquisition through C-Imaging only
- Swapped with OrcaER (uses same CCD sensor chip)
- Run from \\Lister computer (on right)
- Cooled color CCD, MicroPublisher (3.3 mega pixel)
- Acquisition either C-Imaging or QCapture software
Comparison of the Cameras
attached to the Leica Inverted Microscope
|
Camera
Comparison |
 |
 Monochrome Monochrome
|
 Color Color
|
| Camera |
HQ2 |
OrcaER |
MicroPublisher 3.3 (sometimes 5.0) |
| Manufacturer |
Photometrics |
Hamamatsu |
Q-Imaging |
| Optimized for: |
Dim fluorescence &/or quantification of fluorescence
intensity. B/W transmitted light imaging |
Dim fluorescence &/or quantification of fluorescence
intensity. B/W transmitted light imaging |
High resolution transmitted & bright fluorescence |
| Location on microscope |
On left side port |
On left side port |
On top if trinocular |
| Light transmission from microscope |
100% |
100% |
100% or 50% |
| Computer to use |
\\Lister (computer on the right) |
\\Lister (computer on the right) |
\\Snell (computer on the left) |
| Resolution |
Better than presented by 0.63x coupler |
Better than presented by 0.63x coupler |
Better matched to image presented by microscope |
| Preview |
Computer monitor (C-Imaging) |
Computer monitor (C-Imaging) |
Computer monitor (C-Imaging or Q-Capture) |
| Max..field of view size |
1300 by 1024 pixels |
1300 by 1024 pixels |
2048 by 1542 pixels |
| Min. field of view size |
|
Sub region of interest supported |
512 by 385 pixels |
| Intensity depth |
8 or 12 bit B/W |
8 or 12 bit B/W |
8 or 10 bit each RGB channels (24 or 30 bit total) |
| Color balance |
not applicable |
not applicable |
Good after performing auto-balance |
| Sensitivity |
good |
good |
mediocre |
| Far capability |
good |
good |
none |
| Quantification of intensity |
excellent |
excellent |
mediocre |
| Acquisition software |
C-Imaging |
C-Imaging |
C-Imaging or Q-Capture or ImageJ |
| Cooled |
yes |
yes |
no |
| Interface |
Firewire 1394 |
Firewire 1394 |
Firewire 1394 |
| Power up |
Any time, before C-Imaging is run |
Any time, before C-Imaging is run |
Can connect/disconnect with power on |
| Power source |
External Power brick (switched)/ |
External Power supply |
Firewire cable |
Computer:
- Generic PC
- Computer name is \\Lister (\\lister.med.unc.edu) is under the cart on the right side
- The computer is under the cart on the right side
- Other computer name is \\Snell fresnel.med.unc.edu
- Pentium 4, 2.0 GHz, 512 MByte RAM
- 24x CD-RW
- Network
- Name is \\Snell on the MHmicroscopy
domain
- Shared directory on the Windows network is users or users1
or users2 and is
accessible directly from Windows
- Can put \\Snell\users2 in the address box of windows
explorer or MyComputer in order to browse the directory. Enter your
facility username as mhmicroscopy\username
- If you are browsing from Internet Explorer inside UNC try clicking
\\Snell\users1
- This computer is a member of the
MHMICROSCOPY domain
- The MHMICROSCOPY file server is accessible
from this workstations (Snell) computer. Many users' directories are
at \\minsky\users2. This
shared directory can be mapped to the M: drive on your log in.
- Each user requires a username and password on the
MHMICROSCOPY domain in order to log on and access the network
shares
- Software:
- C-Imaging - runs either camera
- Q-Capture - runs cooled CCD color camera only (Micro Publisher, by
Q-Imaging)
- ImageJ - runs the cooled CCD color camera (don't run at the same time
as Q-Capture)
- WinZip - For compressing files and directories into zip
files
- LViewPro - small and quick image viewer for 8 bit b/w and
rgb images
Setting Up the Microscope
- Powering up

- If fluorescence is required switch on Mercury Lamp if it is not already
on (green switch on external power supply). Do not turn the lamp on
unless the lamp housing is cool. (Mercury lamps must be have cooled
for at least 30 minutes before igniting)
- Switch on incandescent lamp for transmitted or DIC (switch is on left
side in front of lamp voltage wheel (1) )

- Switch on Hamamatsu camera control box.
- Switch on computer, if it is not already running.
- Set up optics on the Leica Inverted
- For transmitted light (Kohler Illumination) - if
transmitted not required then go to
fluorescence setup
- Open field aperture (2)
- Swing in neutral density filters (3)
- Minimize condenser aperture (4)
- Set condenser turret ring to H (brightfield)
(5)
- Lower objectives with coarse focus knob (6)
- Select no Wollaston prism - "H" position on wheel below objective
turret (7)
- Choose required objective. (Generally choose a lower power dry
objective, locate sample and region of interest and then move to a higher
power dry or immersion objective )
- Pull out analyzer slider on left side of scope
(8)
- Close B/W camera port by pushing in rod on right hand side of
microscope (9)
- Close color camera port by pushing in rod on left side of the
trinocular head (10)
- Set correction collars on eyepieces to just expose silver ring
(11).
This makes viewing more comfortable for normal eyes and helps keep parfocality with cameras
- Ensure incandescent lamp power is at about 10 - check power wheel by rotating away
until light can be seen spilling onto condenser (12)
- Place sample on stage cover slip down - (This is an inverted
microscope!)
- Focus onto sample
- Close field iris (2) until you see a ring. Focus
the image of the ring with the condenser
focus knob (5), and center it with the 2 knurled screws
at 45 degrees.
- Open field aperture (2) to just fill the field of view seen through the
eye pieces
- Open condenser aperture (4) to desired contrast. Remember maximum
resolution is obtained with a fully open aperture, which also gives
minimum depth of focus & contrast and maximum illumination intensity.
- These steps have set up Kohler Illumination!
- Switch to higher power objective if desired
- For Nomarski (DIC) - if desired
- Set up for Kohler
- Open condenser aperture (4) maximally
- Ensure both Wollaston prisms are not in the light path
(5) and (7)
- Push in analyser
- Adjust upper polariser for maximum darkness
- Select condenser Wollaston which matches objective being used
(5)
- Select Wollaston below object (7) which
matches objective power used (see table on front of scope body)
- Adjust Wollaston (7) prism shear for a
uniform brick gray kind of color by slightly moving the Wollaston wheel
- For fluorescence (* denotes steps also
described in transmitted light setup)
- * Lower objectives
- Pull out analyzer slider on left side of scope
(8)
- Select no Wollaston prism - "H" position on wheel below objective
turret (7)
- * Choose required objective. (Generally choose a lower power,
locate sample and region of interest and then move to a higher power)
- * Close B/W camera port by pushing in rod on right hand side of
microscope
- * Close color camera port by pushing in rod on right side of microscope
and pulling out left side of the trinocular head
- * Set correction collars on eyepieces to just expose silver ring.
This makes viewing more comfortable for normal eyes and helps keep
parfocality with cameras
- Ensure incandescent lamp is off at the switch on the front left of the
scope
- * Place sample on stage cover slip down - (This is an inverted
microscope!)
- Choose filter set using turret just above the left side camera port
- 1=none 2=fitc 3=tritc/texas
red 4=dapi
- Open light path from mercury lamp by pulling rod on back left of scope
out. 1st position out includes an IR filter (don't use) and all the way out
removes the IR filter.
- Focus on sample
- Switch to higher power objective if desired
Image Acquisition
 Acquiring
Images Using SimplePCI <--- please click on this link
Acquiring
Images Using SimplePCI <--- please click on this link
 Acquiring Images Using the
Q-Imaging Software with the MicroPublisher
Color Camera
Acquiring Images Using the
Q-Imaging Software with the MicroPublisher
Color Camera

- Log on the computer
- enter username (and press tab to move to-)
- enter password
- choose domain MHMICROSCOPY (not local computer
Snell)
- Choose QCapture
N.B. clicking on these links should open a view in
another window. This window may be hidden behind other windows on your
computer.
- open control tools (yellow
wrench)
- open live view (green play
symbol)
- In the tools window choose bin 1
- display live image
- white balance
- select 8 bit mode, which is generally adequate and makes viewing image
file easier
- auto expose (due to a software bug always do auto expose with a bin of
1, then switch to another bin size as desired)
- choose resolution (i.e. bin size: 1=2048*1536 2=1024*768
3=768*512 4=512*384)
- snap image
- save image
- repeat ad lib.
- Shutdown
- Turn off cooler by unchecking box in tools window or unplug
Firewire
connector to camera
- Log off
- Lower objective
- Clean up oil on all objectives used with immersion oil
- Power down arc lamp if no one else will be using fluorescence
|
 |
 |
Copyright 2001-2014 Dr. M. Chua,
Program in Molecular Biology & Biotechnology, School of Medicine, University of North Carolina, Chapel Hill, NC 27599
|
| Go Back |
Booking Resources |
Questions/comments/problems:
Michael Chua
|
|
|
 |
Last Updated:
2014-05-14 |
.gif) Leica
DMIRB
Inverted Microscope with Color & B/W Cooled Digital
Cameras
Leica
DMIRB
Inverted Microscope with Color & B/W Cooled Digital
Cameras
.gif)


![]() Acquiring
Images Using SimplePCI <--- please click on this link
Acquiring
Images Using SimplePCI <--- please click on this link ![]() Acquiring Images Using the
Q-Imaging Software with the MicroPublisher
Color Camera
Acquiring Images Using the
Q-Imaging Software with the MicroPublisher
Color Camera
![]()